
New Progress from ECUST in Live-Cell RNA Imaging Published in Nature Protocols
Recently, a research team from the School of Pharmacy, the State Key Laboratory of Bioreactor Engineering, and the Optogenetics and Synthetic Biology Interdisciplinary Research Center at ECUST, led by Professors Yi Yang and Xianjun Chen, made new advances in live-cell RNA imaging. Their work was published in Nature Protocols under the title “Live-cell imaging of RNA dynamics using bright and stable fluorescent RNAs”.
RNAs exhibit complex dynamics in cells, including expression, splicing, localization, translation and degradation, and these processes are highly coordinated and tightly regulated both spatially and temporally. To better understand the biological function of diverse RNAs, approaches that allow monitoring of RNA with high spatiotemporal resolution are essential. Fluorescent RNAs (FRs) have emerged as a promising tool, using RNA aptamers as tags that specifically bind and activate small-molecule dyes to generate fluorescence.
The advantages of this approach include operational simplicity, minimal perturbation, and high signal-to-noise ratio. In previous studies, the team of Professors Yang and Chen developed high-performance FRs, including Pepper, Clivia, and Okra, enabling in situ labeling and dynamic imaging of diverse RNA species in higher eukaryotic cells. (Nature Biotechnology, 2019; Nature Chemical Biology, 2021, 2024a, 2024b; Nature Methods, 2023).
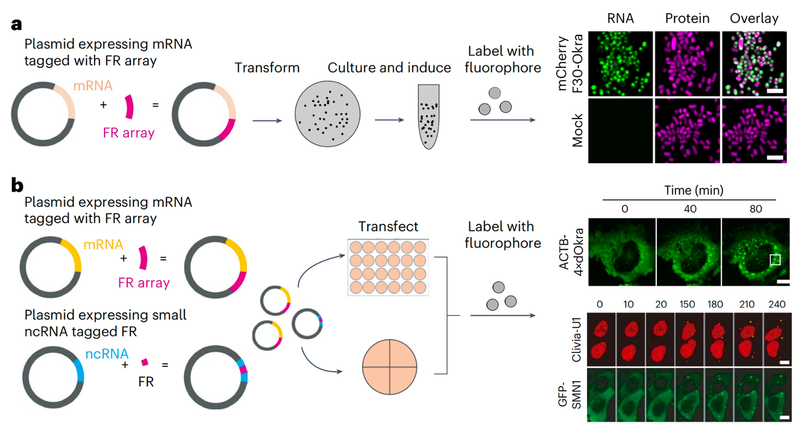
In this study, the team summarized the photophysical and chemical properties of Pepper, Clivia, and Okra RNAs, and constructed a comprehensive live-cell RNA labeling and dynamic imaging platform covering multiple key applications. In addition, the team provided operational guidelines, addressed technical challenges, and offered validation strategies, enhancing the usability and reliability of FR tools. This work has provided a robust methodological foundation and reference for in-depth studies of RNA regulation in physiological and pathological processes.
Drs. Fangting Zuo, Ni Su, and Xin Xie serve as co-first authors of this study. Professors Yi Yang and Xianjun Chen are the corresponding authors. The research was supported by the National Key Research and Development Program of China, the National Natural Science Foundation of China, the Shanghai Frontiers Science Center of Optogenetic Techniques for Cell Metabolism, and the State Key Laboratory of Bioreactor Engineering.